Úvod do Světa Terbutrynu: Identifikace a Základní Charakteristika

V rozsáhlém a neustále se vyvíjejícím světě zemědělských chemikálií zaujímá herbicid terbutryn specifické a významné postavení. Tato organická sloučenina, patřící do skupiny triazinových herbicidů, se etablovala jako účinný nástroj v boji proti širokému spektru nežádoucích rostlin, které mohou negativně ovlivnit výnosy zemědělských plodin. Abychom plně pochopili roli a dopad terbutrynu, je nezbytné se ponořit do jeho základní chemické struktury, fyzikálních a chemických vlastností a mechanismu účinku na cílové rostliny.
Chemická Identifikace Terbutrynu: Na molekulární úrovni je terbutryn charakterizován svou složitou strukturou, která zahrnuje heterocyklický triazinový kruh. Přesná chemická nomenklatura podle IUPAC (Mezinárodní unie pro čistou a aplikovanou chemii) zní 2-(terc-butylamino)-4-(ethylamino)-6-(methylthio)-s-triazin. Tento systematický název poskytuje přesný popis atomového uspořádání a funkčních skupin přítomných v molekule terbutrynu. Sumární chemický vzorec terbutrynu je C₁₀H₁₉N₅S, což kvantitativně vyjadřuje počet a typ atomů tvořících jednu molekulu této látky.
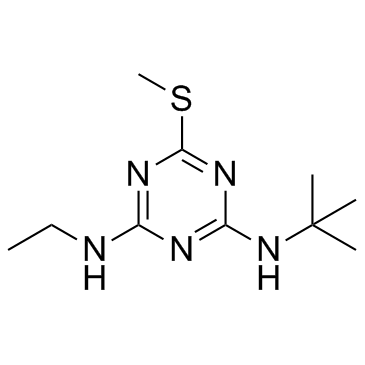
Fyzikální a Chemické Vlastnosti: Terbutryn se za standardních podmínek vyskytuje jako bílá krystalická látka. Jeho molární hmotnost činí přibližně 241,35 g/mol, což je důležitý parametr pro výpočty v chemii a environmentální vědě. Bod tání terbutrynu se pohybuje v rozmezí 104-105 °C, což ovlivňuje jeho stabilitu a chování v různých teplotních podmínkách prostředí. Jedním z klíčových aspektů z hlediska jeho aplikace a environmentálního osudu je jeho rozpustnost ve vodě, která je relativně nízká, činí přibližně 25 mg/l při 20 °C. Tato vlastnost má významný vliv na jeho mobilitu v půdě a potenciál pro kontaminaci vodních zdrojů. Na druhou stranu, terbutryn vykazuje dobrou rozpustnost v organických rozpouštědlech, což je důležité pro formulaci herbicidních přípravků.
Mechanismus Účinku na Rostliny: Terbutryn působí jako selektivní systémový herbicid. To znamená, že je rostlinami absorbován, primárně kořeny a listy, a následně translokován (přesouván) v rámci rostlinného těla. Na buněčné úrovni terbutryn inhibuje fotosyntézu, klíčový biochemický proces, který umožňuje rostlinám přeměňovat světelnou energii na chemickou energii ve formě cukrů. Konkrétně terbutryn blokuje transport elektronů v fotosystému II na úrovni D1 proteinu. Tato inhibice vede k zastavení produkce energie, narušení metabolických procesů a v konečném důsledku k odumření citlivých plevelů. Selektivita terbutrynu, tedy jeho schopnost hubit plevele bez poškození cílové plodiny, je dána rozdíly v absorpci, translokaci a především v metabolismu herbicidu mezi různými rostlinnými druhy. Některé plodiny jsou schopny terbutryn rychleji detoxikovat pomocí enzymatických systémů, čímž se chrání před jeho toxickými účinky.
Pochopení těchto základních aspektů terbutrynu je klíčové pro posouzení jeho přínosů v zemědělství, ale také pro hodnocení potenciálních rizik spojených s jeho používáním pro životní prostředí a lidské zdraví. V následujících částech tohoto článku se budeme podrobněji zabývat specifickými oblastmi použití terbutrynu, jeho vlivem na ekosystémy, regulačními opatřeními a možnými alternativami.
Široké Spektrum Použití Terbutrynu v Moderním Zemědělství
Terbutryn si díky své účinnosti a relativní selektivitě získal významné postavení v moderním zemědělství. Je široce využíván pro kontrolu širokého spektra jednoletých a víceletých plevelů v různých polních plodinách. Jeho aplikace přispívá k zajištění optimálního růstu pěstovaných rostlin, maximalizaci výnosů a usnadnění zemědělských praktik. Mezi klíčové plodiny, u kterých se terbutryn běžně používá, patří kukuřice, slunečnice, cukrová řepa, sója, brambory a některé druhy zeleniny. Účinnost terbutrynu se projevuje jak proti dvouděložným, tak proti některým jednoděložným plevelům, což z něj činí cenný nástroj v integrované ochraně rostlin.
Specifické Aplikace v Různých Plodinách:
- Kukuřice: V pěstování kukuřice se terbutryn často používá preemergentně (před vzejitím plevelů a plodiny) nebo postemergentně (po vzejití). Účinně kontroluje například ježatku kuří nohu, laskavce, merlíky, rdesna a některé trávy. Jeho použití pomáhá zajistit raný růst kukuřice bez konkurence plevelů o živiny, vodu a světlo, což je klíčové pro dosažení vysokých výnosů.
- Slunečnice: Terbutryn je důležitou součástí herbicidních programů i v pěstování slunečnice. Pomáhá v boji proti pcháčům, svlačcům, ambroziím a dalším obtížným plevelům, které mohou výrazně snížit produkci semen a kvalitu oleje. Vzhledem k citlivosti některých odrůd slunečnice je však nutné pečlivě dodržovat doporučené dávkování a aplikační termíny.
- Cukrová Řepa: Pěstování cukrové řepy je náročné na kontrolu plevelů, a terbutryn zde hraje významnou roli, často v kombinaci s dalšími herbicidy v rámci komplexních postřikových programů. Účinně potlačuje merlíky, lebedy, heřmánky, rdesna a další běžné plevele na cukrovkových polích. Správná herbicidní ochrana je zásadní pro dosažení optimálního vývoje bulev a vysokého obsahu cukru.
- Sója: V systémech pěstování sóji se terbutryn používá méně často samostatně a spíše v kombinacích s jinými herbicidy, aby se dosáhlo širšího spektra účinnosti proti plevelům, jako jsou laskavce, svlačce, ambrozie a některé trávy. Volba herbicidního programu v sóji závisí na konkrétním plevelném spektru a pěstované odrůdě.
- Brambory: Terbutryn může být aplikován i v bramborách pro preemergentní kontrolu mnoha dvouděložných plevelů, čímž se snižuje potřeba následných mechanických nebo postemergentních zásahů. Účinná herbicidní ochrana je důležitá pro zajištění dobrého nasazení hlíz a usnadnění sklizně.
- Zelenina: V některých druzích zeleniny, jako je například hrách nebo mrkev, může být terbutryn použit pro preemergentní nebo časně postemergentní kontrolu specifických plevelů. Vždy je však nutné zohlednit citlivost daného druhu zeleniny a dodržovat přesné dávkování a ochranné lhůty.


Formulace a Aplikace: Terbutryn je k dispozici v různých formulacích, včetně suspenzních koncentrátů (SC), dispergovatelných granulí (WG) a dalších. Způsob aplikace se liší v závislosti na plodině, plevelném spektru a podmínkách prostředí. Může být aplikován postřikem na půdu před setím nebo po setí, ale před vzejitím plodiny (preemergentně), nebo postřikem na vzešlé plevele (postemergentně). Dávkování se pečlivě řídí doporučeními výrobce a zohledňuje typ půdy, fázi růstu plodiny a plevelů a další faktory. Moderní zemědělské postřikovače umožňují přesnou aplikaci herbicidů, minimalizaci úletu a optimalizaci pokrytí cílových rostlin.
Přes svou účinnost je však nutné si uvědomit, že nadměrné nebo nesprávné používání terbutrynu může vést k rezistenci plevelů, kdy se populace plevelů stávají méně citlivými nebo zcela odolnými vůči jeho účinku. Proto je v rámci integrované ochrany rostlin (IPM) kladen důraz na střídání herbicidů s různými mechanismy účinku, kombinaci chemických a nechemických metod kontroly plevelů a monitorování citlivosti plevelných populací.

Vliv Terbutrynu na Životní Prostředí: Komplexní Hodnocení Rizik
Používání terbutrynu v zemědělství, podobně jako u mnoha jiných pesticidů, s sebou nese potenciální rizika pro životní prostředí. Důkladné posouzení těchto rizik je nezbytné pro zajištění udržitelného zemědělství a ochrany ekosystémů. Vliv terbutrynu na životní prostředí zahrnuje jeho chování v půdě a vodě, jeho toxicitu pro necílové organismy a potenciál pro bioakumulaci a biomagnifikaci.
Chování Terbutrynu v Půdě a Vodě: Po aplikaci na pole se terbutryn může dostat do různých složek životního prostředí. Jeho osud v půdě je ovlivněn několika procesy, včetně adsorpce na půdní částice (zejména organickou hmotu a jílové minerály), mikrobiálního rozkladu, chemické hydrolýzy a fotodegradace. Míra těchto procesů závisí na faktorech, jako je typ půdy, vlhkost, teplota a mikrobiální aktivita. Relativně nízká rozpustnost terbutrynu ve vodě sice omezuje jeho okamžitou mobilitu, ale srážky a zavlažování mohou vést k jeho splachování z povrchu půdy do vodních toků nebo k prosakování do podzemních vod. Kontaminace povrchových a podzemních vod terbutrynem je významným environmentálním problémem, neboť může ohrozit vodní ekosystémy a zdroje pitné vody.
Toxický Vliv na Vodní Organismy: Terbutryn je známý svou vysokou toxicitou pro některé vodní organismy, zejména pro řasy a sinice (cyanobakterie). Jako inhibitor fotosyntézy narušuje jejich základní životní procesy, což může vést k poklesu jejich populací a následně k narušení celých vodních potravních řetězců. Studie prokázaly, že i nízké koncentrace terbutrynu mohou mít negativní vliv na růst a fyziologii těchto primárních producentů. Kromě řas může být terbutryn toxický i pro některé vodní bezobratlé (např. dafnie) a ryby, i když jeho toxicita pro tyto skupiny organismů je obecně nižší než pro řasy. Dlouhodobé expozice nízkým koncentracím však mohou mít chronické účinky na jejich reprodukci a vývoj.
Vliv na Necílové Terestrické Organismy: Kromě vodních organismů je důležité posoudit i vliv terbutrynu na necílové terestrické organismy, jako jsou hmyz (včetně opylovačů, jako jsou včely), ptáci a půdní mikroorganismy. Obecně se má za to, že terbutryn má relativně nízkou akutní toxicitu pro včely, pokud je aplikován v souladu s doporučenými postupy a mimo období kvetení. Nicméně, potenciální subletální účinky na jejich chování a imunitní systém nelze zcela vyloučit a vyžadují další výzkum. Podobně, přímá toxicita terbutrynu pro ptáky a savce je považována za nízkou při běžných aplikačních dávkách. Nicméně, nepřímé účinky, například snížení dostupnosti potravy v důsledku hubení plevelů, mohou mít negativní dopady na jejich populace. Vliv terbutrynu na půdní mikroorganismy je komplexnější. Některé studie naznačují, že vysoké koncentrace mohou dočasně narušit jejich aktivitu a diverzitu, což může mít vliv na koloběh živin v půdě. Při běžných zemědělských dávkách jsou však tyto účinky obvykle považovány za přechodné a relativně malé.
Potenciál pro Bioakumulaci a Biomagnifikaci: Bioakumulace je proces, při kterém se chemická látka hromadí v těle organismu rychleji, než je vylučována. Biomagnifikace je pak proces, při kterém se koncentrace chemické látky zvyšuje s postupem po potravním řetězci. V případě terbutrynu existují určité důkazy o jeho schopnosti bioakumulovat v některých vodních organismech, zejména v řasách a rybách. Nicméně, jeho potenciál pro biomagnifikaci v celých potravních řetězcích se považuje za relativně nízký ve srovnání s některými persistentními organickými polutanty (POPs). Přesto je důležité monitorovat jeho přítomnost v různých složkách životního prostředí a potravních řetězcích, aby se předešlo potenciálním dlouhodobým rizikům.
Environmentální Monitoring a Výzkum: Pro lepší pochopení a minimalizaci environmentálních rizik spojených s používáním terbutrynu je nezbytný systematický monitoring jeho přítomnosti v půdě, vodě a biote. Pravidelné analýzy vzorků z různých lokalit mohou poskytnout cenné informace o jeho koncentracích, šíření a trendech v čase. Dále je důležitý kontinuální výzkum zaměřený na detailní studium jeho chování v různých typech ekosystémů, jeho toxicity pro široké spektrum organismů a potenciálních synergických efektů s jinými znečišťujícími látkami. Tyto